Innovative Drug Research Solutions
About Us
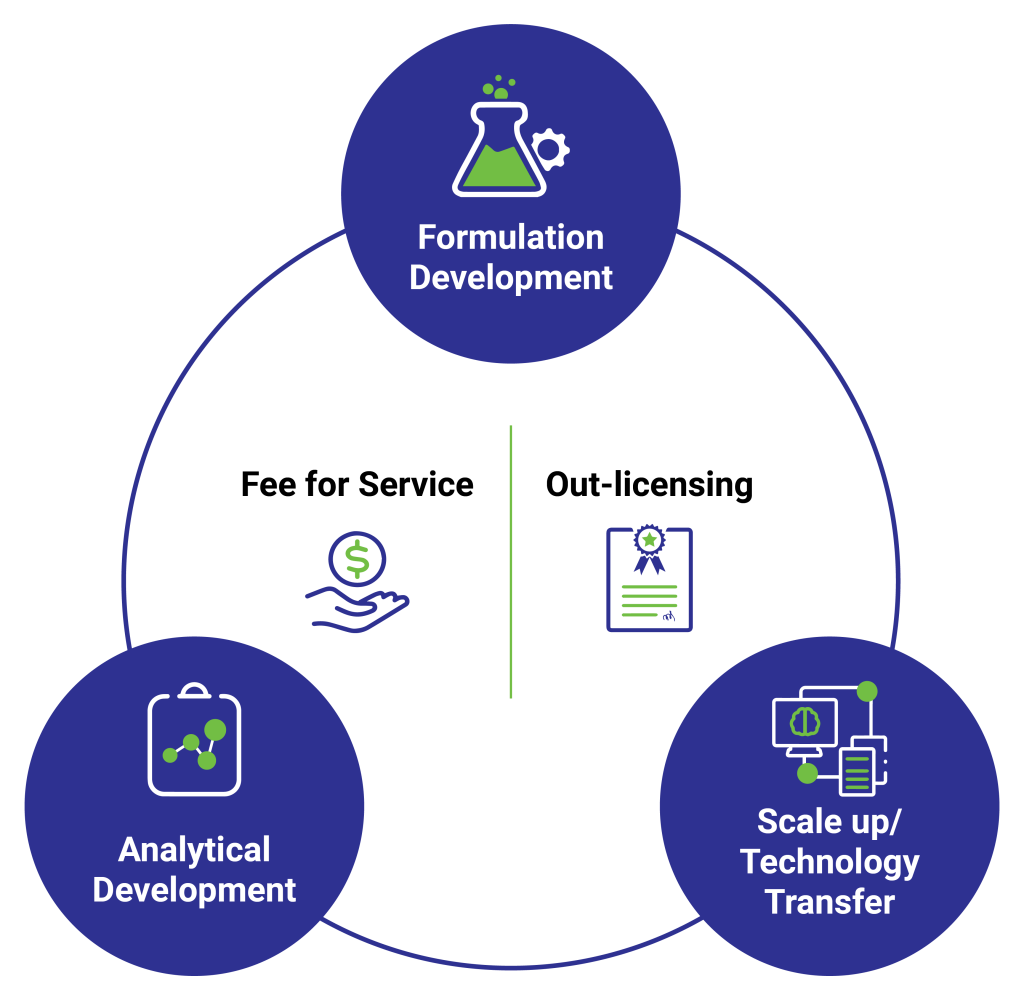
IDRS Labs – a specialty pharmaceutical organization, is a leading product development partner to pharmaceutical organizations globally. IDRS has a proven track record across domains, forging and nurturing strategic, long-term partnerships with pharmaceutical leaders, with a goal of enhancing therapeutic value of products for consumers, and building success for stakeholders. We have helped our clients develop products – from injectables to orals, which have been a commercial success in global markets, and are a testimony to our expertise and experience as a research, development and growth partner.
MANAGEMENT
 Shivkumar, a trained Clinical Pharmacist, brings significant experience in core clinical research. He has been involved in conducting multiple clinical trials in various indications for India and global registrations. He has experience in conducting Phase-II to IV clinical trials for small molecules and bio-similars.
Shivkumar, a trained Clinical Pharmacist, brings significant experience in core clinical research. He has been involved in conducting multiple clinical trials in various indications for India and global registrations. He has experience in conducting Phase-II to IV clinical trials for small molecules and bio-similars.
Read more..
 Mahendra Joshi brings significant experience in solid and liquid oral formulation development. He has experience in setting up the formulation laboratory and in establishing the systems for preformulation and NCE product development. He has successfully developed drug products for global pharma majors to support IND, CTA, NDA and ANDA fillings.
Mahendra Joshi brings significant experience in solid and liquid oral formulation development. He has experience in setting up the formulation laboratory and in establishing the systems for preformulation and NCE product development. He has successfully developed drug products for global pharma majors to support IND, CTA, NDA and ANDA fillings.
Read more..
 Yogesh is an entrepreneur who has been a part of many successful ventures in the pharmaceutical industry. He brings substantial experience to IDRS in areas of Finance, Business Strategy and Operations (including logistics and supply chain management).
Yogesh is an entrepreneur who has been a part of many successful ventures in the pharmaceutical industry. He brings substantial experience to IDRS in areas of Finance, Business Strategy and Operations (including logistics and supply chain management).
Read more..